Online Database of Chemicals from Around the World
| Tianjin J & L Forever Trading Corp. | China | |||
|---|---|---|---|---|
 | www.jlforever.com | |||
 | +86 (22) 6539-0499 6622-0951 6628-0372 | |||
 | +86 (22) 6622-0967 | |||
 | sales@tjril.com goldenstar@china.com | |||
| Chemical distributor since 1994 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Shijiazhuang Lemandou Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.lemandou.com | |||
 | +86 (311) 8778-7400 | |||
 | +86 (311) 8778-7409 | |||
 | sales@lemandou.com lemandou@hotmail.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Chaitanya Chemicals | India | |||
|---|---|---|---|---|
 | www.bariumindia.com | |||
 | +91 (8562) 243-179 | |||
 | +91 (8562) 243-179 | |||
 | info@bariumindia.com | |||
| Chemical manufacturer since 1989 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Tianjin Minle Chemical Import & Export Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tjminlechem.com | |||
 | +86 (22) 8425-9028 | |||
 | eric@tjminlechem.com | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Zhushan County Qinba Barium Salt Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.longfu-group.com | |||
 | +86 (27) 8557-8968 | |||
 | cherry@longfu-group.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shandong Qishun Chemical Trade Co., Ltd | China | |||
|---|---|---|---|---|
 | www.qishunchem.cn | |||
 | +8618954925963 | |||
 | qishunchem@gmail.com | |||
| Chemical distributor since 2018 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Shandong S Sailing Chemical Co., Ltd | China | |||
|---|---|---|---|---|
 | www.ssailing.cn | |||
 | +86 13325053493 | |||
 | qihang@ssailing.cn | |||
| Chemical distributor since 2012 | ||||
| chemBlink Standard supplier since 2021 | ||||
| Shandong Near Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.nearchemical.net | |||
 | +86 15615461505 | |||
 | belinda@nearchemical.net | |||
 | QQ Chat | |||
| Chemical distributor since 2012 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Shandong Xuke Chenmical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xukechem.com | |||
 | +86 18554324858 | |||
 | myla@xukechem.com | |||
| Chemical manufacturer since 2018 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Riverland Trading LLC. | USA | |||
|---|---|---|---|---|
 | www.riverlandtrading.com | |||
 | +1 (336).944-2293 | |||
 | bens@riverlandtrading.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2023 | ||||
| Weifang Xinruida Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xrdchemical.com | |||
 | +86-536-8953288 | |||
 | info@xrdchemical.com | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Jointech Development Co., Ltd. | China | |||
|---|---|---|---|---|
 | jointechem.com | |||
 | +86 17802201425 | |||
 | service@jointechem.com | |||
| Chemical distributor since 2021 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Thermo Fisher Scientific | USA | |||
|---|---|---|---|---|
 | www.fishersci.com | |||
 | +1 (800) 766-7000 | |||
 | +1 (800) 926-1166 | |||
 | fs.order@thermofisher.com | |||
| Chemical manufacturer | ||||
| Boya (Tianjin) International Limited | China | |||
|---|---|---|---|---|
 | www.boya-chemical.com | |||
 | +86 (22) 2430-2708 2430-2718 | |||
 | +86 (22) 2430-2728 / 2430-2738 | |||
 | boya.chemical@163.com | |||
| Chemical distributor | ||||
| Avonchem/Chromos Express Ltd. | UK | |||
|---|---|---|---|---|
 | www.avonchem.co.uk | |||
 | +44 (1625) 434-300 | |||
 | +44 (1625) 869-777 | |||
 | info@avonchem.co.uk | |||
| Chemical manufacturer | ||||
| Hebei Medipharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hbmedipharm.com | |||
 | +86 (311) 8613-6565 8603-2913 | |||
 | +86 (311) 8696-8724 / 8698-5695 | |||
 | sales@hbmedipharm.com | |||
| Chemical manufacturer since 2004 | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| Classification | Inorganic chemical industry >> Inorganic salt >> Metal sulfides and sulfates |
|---|---|
| Name | Sodium sulfide |
| Molecular Structure | 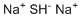 |
| Molecular Formula | Na2S |
| Molecular Weight | 78.03 |
| CAS Registry Number | 1313-82-2 |
| EC Number | 215-211-5 |
| SMILES | [Na+].[Na+].[SH-] |
| Density | 1.856 g/mL (Expl.) |
|---|---|
| Melting point | 950 °C (Expl.) |
| Solubility | water 186 g/L (20 °C), soluble (alcohol (Expl.) |
| Hazard Symbols |     GHS05;GHS06;GHS07;GHS09 Danger Details GHS05;GHS06;GHS07;GHS09 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H290-H301+H311-H301-H302-H311-H314-H318-H400 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P234-P260-P262-P264-P264+P265-P270-P273-P280-P301+P316-P301+P317-P301+P330+P331-P302+P352-P302+P361+P354-P304+P340-P305+P354+P338-P316-P317-P321-P330-P361+P364-P363-P390-P391-P405-P406-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 1385 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Sodium sulfide is an inorganic compound with the chemical formula Na2S. It exists as a colorless to yellowish solid and is highly water-soluble, forming strongly alkaline solutions that release hydrogen sulfide (H2S) gas upon exposure to acids. Sodium sulfide has been widely used in industrial processes since the 19th century and remains important in various applications such as chemical manufacturing, leather processing, and water treatment. The compound is most commonly produced by the direct reduction of sodium sulfate (Na2SO4) with carbon at elevated temperatures, typically in a rotary furnace. Another common method involves reacting sulfur with sodium hydroxide (NaOH) or sodium metal in an aqueous or molten state. Commercial sodium sulfide is often encountered as a hydrated solid or in solution form, depending on the method of storage and use. Historically, sodium sulfide played a key role in the development of the sulfide-based tanning process in the leather industry. Its ability to break down keratin in animal hides makes it useful for unhairing and dehairing during leather preparation. The compound disrupts disulfide bonds in keratin, facilitating the removal of hair from hides before further tanning processes. This application continues to be one of the primary industrial uses of sodium sulfide today. In the pulp and paper industry, sodium sulfide is a critical component of the Kraft process, which is used to produce wood pulp. In this context, sodium sulfide, in combination with sodium hydroxide, helps break down lignin—the binding component in wood—while preserving the cellulose fibers necessary for paper production. The Kraft process revolutionized paper manufacturing in the early 20th century by improving both yield and product quality. Sodium sulfide also finds use as a reducing agent in various chemical syntheses. It is used in the dye and textile industries for the reduction of certain vat dyes and sulfur dyes, and in organic synthesis to introduce sulfide or thiol functionalities. In water treatment, it can serve as an oxygen scavenger and a precipitating agent for heavy metals, forming insoluble metal sulfides that can be removed from wastewater. Due to its strong alkaline nature and its tendency to hydrolyze in water, sodium sulfide must be handled carefully. It reacts with moisture and carbon dioxide in air, producing corrosive and odorous hydrogen sulfide gas. This gas is toxic, flammable, and has a characteristic "rotten egg" odor detectable at very low concentrations. Prolonged exposure or inadequate ventilation can pose significant health risks. For this reason, storage and handling must include safeguards such as sealed containers, ventilation systems, and appropriate personal protective equipment. In addition to its industrial uses, sodium sulfide has applications in laboratory settings as a reagent in inorganic and analytical chemistry. It is used to precipitate metal ions from solution and to prepare metal sulfide nanoparticles. Some specialized applications include its use in photographic processing and in the manufacture of certain rubber chemicals. In summary, sodium sulfide is a versatile inorganic chemical with a long history of use in leather processing, pulping, dyeing, water treatment, and chemical synthesis. Its chemical properties—strong alkalinity, reactivity with acids, and reducing capability—have made it valuable across a range of industrial sectors. However, its reactivity and potential hazards require careful handling and environmental management to ensure safe and effective use. References 2024. Development of a reusable Fe2O3-PANI/CdS nanocomposite for efficient and sustainable treatment of malachite green dye. Journal of Materials Science: Materials in Electronics, 35. DOI: 10.1007/s10854-024-13996-1 2020. Development of dual function polyamine-functionalized carbon dots derived from one step green synthesis for quantitation of Cu2+ and S2− ions in complicated matrices with high selectivity. Analytical and Bioanalytical Chemistry, 412(4). DOI: 10.1007/s00216-019-02362-4 1987. Manipulation of End‐Product Distribution in Strict Anaerobes. Annals of the New York Academy of Sciences, 506(1). DOI: 10.1111/j.1749-6632.1987.tb23811.x |
| Market Analysis Reports |