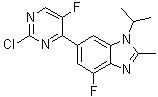
6-(2-Chloro-5-fluoro-4-pyrimidinyl)-4-fluoro-2-methyl-1-(1-methylethyl)-1H-benzimidazole is a heterocyclic compound that combines a substituted benzimidazole core with a pyrimidine moiety. Benzimidazole derivatives have long been recognized in medicinal chemistry for their stability, planarity, and ability to interact with biological targets, while pyrimidine rings are a common motif in nucleobases and biologically active molecules. The fusion of these two structural motifs in a single molecule allows the compound to serve as a scaffold in drug discovery, particularly in areas involving kinase inhibition and receptor modulation.
Structurally, the compound features a benzimidazole core substituted at the 2-position with a methyl group and at the N1 nitrogen with an isopropyl group. The 4-position carries a fluorine atom, and the 6-position is substituted with a pyrimidinyl group that itself bears chloro and fluoro substituents. The combination of halogen substituents and alkyl groups modulates electronic density, lipophilicity, and steric effects, which can influence molecular recognition and binding interactions with enzymes or receptors. The pyrimidine moiety, attached at the 6-position, extends the aromatic system and provides additional hydrogen bonding and π–π stacking capabilities.
The synthesis of such molecules typically involves construction of the benzimidazole ring through condensation of a suitably substituted o-phenylenediamine with a carbonyl compound. N-alkylation at the N1 position introduces the isopropyl group, while selective halogenation at the 4-position installs the fluorine atom. The pyrimidine unit is commonly coupled to the benzimidazole through palladium-catalyzed cross-coupling reactions or nucleophilic aromatic substitution, allowing precise control over regioselectivity and maintaining the integrity of the heterocyclic cores. Substituents on the pyrimidine, such as chloro and fluoro groups, can further influence chemical reactivity and biological properties.
Compounds combining benzimidazole and pyrimidine motifs are widely explored in medicinal chemistry due to their potential as kinase inhibitors. The fused heterocyclic system can fit into ATP-binding pockets of kinases, and the substituents provide opportunities for specific interactions with amino acid residues, enhancing both potency and selectivity. The presence of halogen atoms often improves metabolic stability and can increase binding affinity through halogen bonding or by influencing electronic properties of the heterocycles.
Beyond kinase inhibition, these structural features make the compound a useful scaffold for designing molecules that interact with other biological targets, including nucleic acid-processing enzymes and protein receptors. The extended aromatic system, combined with functionalized heterocycles, provides opportunities for π-stacking, hydrogen bonding, and van der Waals interactions, which are essential in drug-target recognition. Medicinal chemists can use such scaffolds to explore structure–activity relationships, systematically varying substituents to optimize potency, selectivity, and pharmacokinetic properties.
In addition to biological applications, the molecule also has value in synthetic organic chemistry. The benzimidazole-pyrimidine framework is chemically robust yet amenable to further functionalization. Positions on both rings can be selectively substituted through modern cross-coupling reactions, halogenation, or alkylation, making it a versatile intermediate for constructing more complex molecules. The electronic and steric characteristics imparted by the methyl, isopropyl, fluoro, and chloro substituents provide chemists with additional handles for fine-tuning reactivity.
Overall, 6-(2-chloro-5-fluoro-4-pyrimidinyl)-4-fluoro-2-methyl-1-(1-methylethyl)-1H-benzimidazole represents a highly functionalized heterocyclic scaffold that integrates the pharmacological potential of benzimidazole and pyrimidine. Its combination of structural rigidity, aromaticity, halogenation, and alkyl substitution makes it valuable in drug discovery, kinase inhibition studies, and synthetic organic chemistry, illustrating the strategic design of heterocyclic molecules for biological and chemical applications.
References
2018. Abemaciclib. Pharmaceutical Substances.
PubChem Literature ID: 906196375
|





















 GHS07 Warning Details
GHS07 Warning Details