Online Database of Chemicals from Around the World
| Hangzhou StarShine Pharmaceutical Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 | www.starshinepharm.com | |||
 | +86 (571) 8512-3681 +86 13777804878 | |||
 | +86 (571) 8512-2157 | |||
 | sales@starshinepharm.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Beijing Huikang Boyuan Chemical Tech Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 | www.huikangchem.com | |||
 | +86 (10) 6886-2197 6886-7502 | |||
 | +86 (10) 6888-2204 | |||
 | market@huikangchem.com sales@huikangchem.com sales3@huikangchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Taizhou Crene Biotechnology Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| ZHIYU Biotechnology Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 | www.zhiyubiotech.com | |||
 | +86 (512) 6279-1916 | |||
 | +86 (512) 6279-1915 | |||
 | sales@zhiyubiotech.com info@zhiyubiotech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Changzhou Carbochem Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 | www.carbo-chem.com | |||
 | +86 (519) 8918-1862 +86 13775204319 | |||
 | +86 (519) 8918-1862 | |||
 | gao@carbo-chem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | Inquire | ||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Zhejiang Warrant Pharmaceutical Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 | www.zhejiang-warrant.com | |||
 | +86 (512) 8518-0611 +86 17312581805 | |||
 | +86 (512) 8917-1181 | |||
 | sales@zhejiang-warrant.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Xiamen Halosyntech Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 | www.halosyntech.com | |||
 | +86 (592) 668-8068 | |||
 | +86 (592) 666-8067 | |||
 | admin@halosyntech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Qichuang Chemical Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 | www.qc-chemical.com | |||
 | +86 (571) 8893-5129 | |||
 | +86 (571) 8825-0182 | |||
 | david@qc-chemical.com sales@qc-chemical.com davidw0828@gmail.com | |||
 | QQ Chat | |||
| Chemical distributor since 2009 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Cheminspire Technology Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 | www.cheminspire.com | |||
 | +86 (571) 8908-1561 | |||
 | +86 (571) 8908-1566 | |||
 | info@cheminspire.com | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Chemtech Inductry Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 | www.zjchemtech.com | |||
 | +86 (571) 5671-5668 | |||
 | +86 (571) 85423167 | |||
 | info@zjchemtech.com | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Changzhou Yongxu Chemical Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 | www.czyxchem.com | |||
 | +86 (519) 8528-6591 8561-5661 | |||
 | +86 (519) 8526-6592 | |||
 | export@czyxchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Changzhou Yuanda Pharmaceutical Chemical Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 | www.ydpharm.com | |||
 | +86 (519) 8398-4084 +86 13776846168 | |||
 | +86 (519) 8398-4085 | |||
 | sales@ydpharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Cherry Pharmaceutical Technology Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 | www.cherrypharmatech.com | |||
 | +86 (571) 8163-6070 +86 18042403330 | |||
 | +86 (571) 8163-6070 | |||
 | info@cherrypharmatech.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Cangzhou Enke Pharma-tech Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 | www.enkepharma.com | |||
 | +86 (317) 510-5699 510-6597 +86 15533709196 | |||
 | +86 (317) 510-6596 | |||
 | sale@enkepharma.com enkepharma@126.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: ymzhao | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Suli Pharmaceutical Technology Jiangyin Co., Ltd., | China | Inquire | ||
|---|---|---|---|---|
 | www.sulipharma.com | |||
 | +86 (21) 5197-2823 ex 8033 | |||
 | qian.wang@suli.com | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Health Biochem Technology Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 | www.health-biochem.com | |||
 | +86 (29) 6259-9512 | |||
 | sales@health-biochem.com | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Suzhou Keyuan Pharmaceutical Technology Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 | www.kyyykj.com | |||
 | +86 13656237714 | |||
 | xiaokunfu@kyyykj.com | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Santa Cruz Biotechnology, Inc. | USA | Inquire | ||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| AK Scientific, Inc | USA | Inquire | ||
|---|---|---|---|---|
 | www.aksci.com | |||
 | +1 (510) 429-8835 | |||
 | +1 (510) 429-8836 | |||
 | sales@aksci.com | |||
| Chemical manufacturer | ||||
| Classification | Biochemical >> Inhibitor >> Proteases >> HCV protease inhibitor |
|---|---|
| Name | Ledipasvir |
| Synonyms | GS 588 |
| Molecular Structure | 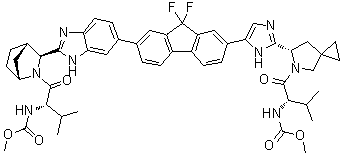 |
| Molecular Formula | C49H54F2N8O6 |
| Molecular Weight | 889.00 |
| CAS Registry Number | 1256388-51-8 |
| EC Number | 694-897-1 |
| SMILES | CC(C)[C@@H](C(=O)N1CC2(CC2)C[C@H]1C3=NC=C(N3)C4=CC5=C(C=C4)C6=C(C5(F)F)C=C(C=C6)C7=CC8=C(C=C7)N=C(N8)[C@@H]9[C@H]1CC[C@H](C1)N9C(=O)[C@H](C(C)C)NC(=O)OC)NC(=O)OC |
| Solubility | Insoluble (5.9E-5 g/L) (25 °C), Calc.*, soluble 20mM (DMSO) |
|---|---|
| Density | 1.42±0.1 g/cm3 (20 °C 760 Torr), Calc.* |
| Index of Refraction | 1.677, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS087 Warning Details GHS087 Warning Details |
|---|---|
| Risk Statements | H371 Details |
| Safety Statements | P260-P264-P270-P308+P316-P405-P501 Details |
| SDS | Available |
|
Ledipasvir is an antiviral drug used primarily for the treatment of hepatitis C virus (HCV) infection. It is part of a class of medications known as direct-acting antivirals (DAAs), which target specific viral proteins to inhibit the replication of HCV. The discovery of ledipasvir arose from extensive research into viral nonstructural proteins crucial for HCV replication, specifically the NS5A protein. This protein plays an essential role in viral RNA replication and assembly, making it a strategic target in antiviral drug development. Ledipasvir was developed by the pharmaceutical company Gilead Sciences and was approved by the FDA in 2014 as part of the combination therapy Harvoni, which includes sofosbuvir, another antiviral drug. The chemical structure of ledipasvir is complex, containing multiple aromatic rings, a fluorinated quinoline, and several nitrogen-containing heterocycles that allow it to interact with and inhibit the NS5A protein. Ledipasvir’s inhibitory action prevents the formation of the HCV replication complex, halting the virus’s ability to reproduce within liver cells. This mechanism has made ledipasvir effective across multiple HCV genotypes, though it is most commonly used to treat genotypes 1 and 4. By targeting NS5A, ledipasvir exhibits a high barrier to resistance, which is an important factor in managing chronic HCV infections. Ledipasvir is administered as an oral tablet in combination with sofosbuvir. The ledipasvir-sofosbuvir combination offers a significant advantage over earlier HCV treatments because it does not require co-administration with interferon or ribavirin, both of which are associated with severe side effects. This combination treatment, known as Harvoni, has a high cure rate, with clinical trials showing that over 90% of patients achieve a sustained virologic response, indicating effective viral eradication. Harvoni is typically taken once daily for 8 to 24 weeks, depending on the severity of the infection, liver function, and prior treatment history. The discovery of ledipasvir has marked a transformative advance in the treatment of hepatitis C. By allowing for a shorter, interferon-free regimen with fewer side effects, it has improved the quality of life for patients and increased the success rate of HCV therapy. Ledipasvir and similar DAAs represent a new era in antiviral therapy, setting the stage for further innovation in the treatment of viral diseases. References 2017. Efficacy and safety of ledipasvir/sofosbuvir with ribavirin in chronic hepatitis C patients who failed daclatasvir/asunaprevir therapy: pilot study. Journal of Gastroenterology. DOI: 10.1007/s00535-017-1380-8 2019. Direct-acting antiviral agents do not increase the incidence of hepatocellular carcinoma development: a prospective, multicenter study. Hepatology International. DOI: 10.1007/s12072-019-09939-2 2021. Comparison of the Prognosis of Decompensated Cirrhosis in Patients with and Without Eradication of Hepatitis C Virus. Infectious Diseases and Therapy. DOI: 10.1007/s40121-021-00441-7 |
| Market Analysis Reports |
| List of Reports Available for Ledipasvir |